Principal Investigator: Antoine LUTTI
Overview
Magnetic Resonance Imaging (MRI) is a powerful non-invasive imaging technique with wide applications in fundamental and clinical neuroscience. The main research interests of the physics group at LREN lie in the development of novel structural, functional and diffusion MRI acquisitions that allow for the characterization of brain structure and the study of its relationship with brain function and behaviour.
Research areas
Quantitative MRI
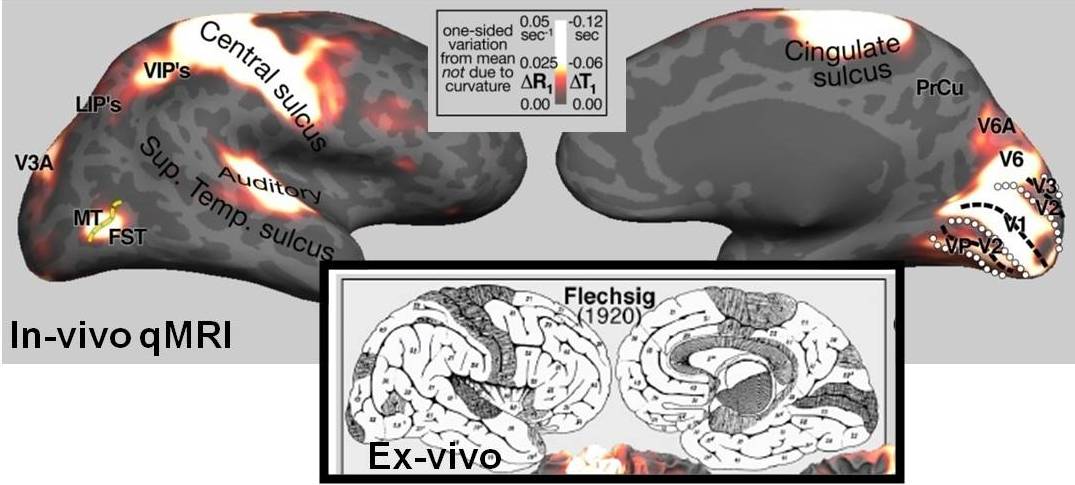
Using MRI techniques developed in-house, we have demonstrated the striking resemblance between quantitative MRI data acquired in-vivo and variations in intra-cortical myelination observed in ex-vivo histological data (Lutti et al., 2014). This demonstrates the ability of qMRI to detect subtle changes in myelination, opening the way for the in-vivo study of the mechanisms underlying myelination changes in health (e.g. plasticity) and disease (e.g. Multiple Sclerosis). We have recently reported the existence of networks of synchronous myelination changes across the cortex (Melie-Garcia et al., 2017). Ongoing work involves the development of novel qMRI biomarkers of tissue microstructure with enhanced specificity (Lutti, ISMRM 2016).
The variations in the MR parameter R1 across the cortex resemble closely myelin density distributions known from ex-vivo histological studies (Lutti et al. 2014).
Prospective correction of patient motion
Head movement leads to severe artefacts in MR images and represents a major hindrance for the use of MRI on clinical populations for the study of brain disease. The laboratory is equipped with state-of-the-art technology to correct for head movement. This technology acts prospectively during image acquisition, by adjusting in real-time the MRI scanner to account for the movement (Zaitsev 2006). While significant improvements in data quality are observed with this technique, strong motion artefacts may remain in the data in non-compliant populations.
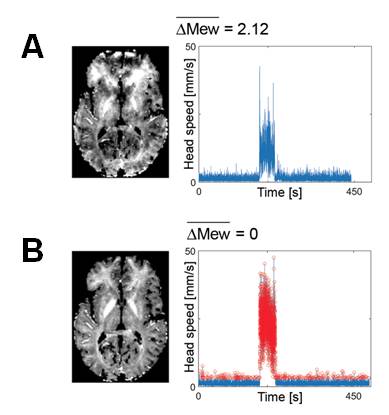
To address this limitation, we have developed a framework that suspends data acquisition during periods of strong motion (Castella 2018), leading to clear improvements in data quality. Head motion is tolerated during innocuous periods of data acquisition, leading to minimal increase in acquisition time due to the suspension. The suspension can be tuned by the user according to the desired image quality and participant’s tolerability.
MR images acquired with prospective motion correction and without (A) and with (B) suspension of data acquisition during periods of head motion. In B, the points of suspended acquisition are highlighted in red. Taken from Castella 2018..
Functional MRI
We have also developed highly optimized 3D EPI acquisitions for fMRI. We have demonstrated a 30% to 60% increase in BOLD sensitivity with 3D EPI at 1.5mm3 resolution at 3T compared to standard 2D EPI sequences (Lutti 2013b). 3D EPI sequences also allow for ultra-fast fMRI acquisitions. For example, whole-brain 3mm3-resolution images can be acquired with a TR of 1s with minimal reduction in signal stability.
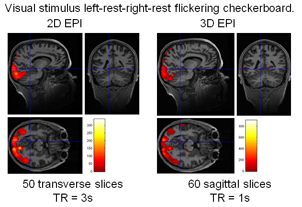
Functional activation detected using a standard 2D EPI sequence (TR=3s) and an ultra-fast 3D EPI sequence (TR=1s) at 3mm3 resolution. Stronger activation is obtained with 3D EPI despite the increased acquisition speed. Taken from Lutti 2013.
Key publications
A complete list of publications is available here.
† Shared senior authorships
Lutti A., Dick F., Sereno M. I., Weiskopf N, 2014.Using high-resolution quantitative mapping of R1 as an index of cortical myelination. Neuroimage, doi: 10.1016/j.neuroimage.2013.06.005.
Sereno, M.I., Lutti A., Weiskopf, N., Dick, F., 2013. In-vivo parcellation of the human cortical surface using quantitative T1 mapping and retinotopy. Cerebral Cortex Sep;23(9):2261-8. doi: 10.1093/cercor/bhs213.
Lutti A., Stadler J., Josephs O., Speck O., Bernarding J., Hutton C., Weiskopf N., 2012. Robust and Fast Whole Brain Mapping of the RF transmit field B1 at 7T. PLoS ONE, 59(3).
Lutti A., Hutton C., Finsterbusch J., Helms G., Weiskopf N., 2010. Optimization and validation of methods for mapping of the radiofrequency field B1 at 3T. Magnetic Resonance in Medicine, 64, 229-238.
Melie-Garcia L, Slater D, Ruef A, Sanabria-Diaz G, Kherif F, Draganski B†, Lutti A†. Networks of Myelin covariance. Human Brain Mapping (2017). doi: 10.1002/hbm.23929
Lorio S, Kherif F, Ruef A, Melie-Garcia L, Frackowiak RS, Ashburner J, Helms G, Lutti A†, Draganski B†. Neurobiological origin of spurious brain morphological changes – a quantitative MRI study. Human Brain Mapping (2016). doi: 10.1002/hbm.23137.
Lutti A., Thomas D.L., Hutton C., Weiskopf N., 2013. High resolution functional MRI at 3T: 3D/2D echo-planar imaging (EPI) with optimized physiological correction. Magn Reson Med. 69(6), 1657-64.
Castella R, Arn L, Dupuis E, Callaghan MF, Draganski B, Lutti A. Controlling for motion artefact levels in prospectively motion corrected MRI data. In revision. Available on BioRxiv: https://doi.org/10.1101/230490


