Aging and increased life expectancy are associated with the progression of chronic metabolic diseases, such as type 2 diabetes, obesity, hypertension, dyslipidemia and cardiovascular conditions. These multifactorial pathologies are complex, often resulting from a combination of genetic and environmental factors, such as physical inactivity and diet. The steady decline of skeletal muscle mass that comes with aging (sarcopenia) is thought to be involved in these metabolic changes, however little is known on how deranged skeletal muscle metabolism influences the development of metabolic disorders.
Pathophysiological framework of skeletal muscle metabolism in aging
The Aging and Muscle Metabolism lab performs translational research projects covering multiple metabolic diseases in human and non-human models, to elucidate molecular mechanisms underlying altered muscle metabolism with aging.
The understanding of the effect of aging and exercise on muscle metabolism is the main theme of our lab. To respond to this quest, we progress through three synchronous aims, which taken together allow an integrative vision.
Schematic representation of our research objectives
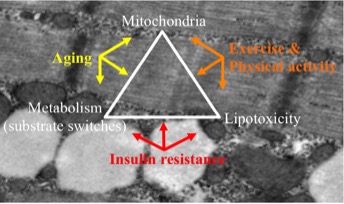
AGING & MUSCLE METABOLISM
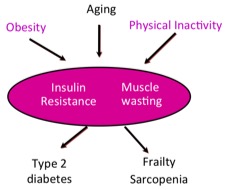
Aging is associated with alterations in skeletal muscle metabolism. In addition to functional consequences, such as muscle wasting, higher risk of falls and reduced independence for daily living, deranged skeletal muscle metabolism is thought to play a role in age-related metabolic diseases, such as type 2 diabetes and obesity.
Healthy aging encompasses multiple interventions and lifestyle changes, such as eating well, maintaining a social role and exercising regularly. Exercise and regular physical activity have been shown to provide many health benefits, particularly in preventing frequent debilitating diseases such as sarcopenia (i.e. marked loss of skeletal muscle mass) and increasing health quality in addition to lifespan. Exercise markedly improves muscle mass and is thought to reverse, at least partially, metabolic consequences of aging.
The Aging and Muscle Metabolism lab uses clinical interventions, such as exercise programs, to understand mechanisms leading to metabolic consequences of aging and to assess reversibility. Participants in our clinical study encompass active or sedentary adults.
 |
 |
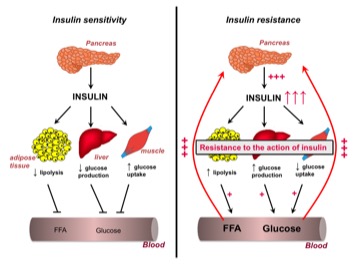
INSULIN RESISTANCE
Insulin resistance can be seen as a state in which a cell, an organ, or the whole body, do not respond to normal amounts of insulin and thus requires higher than average levels of circulating insulin to elicit its normal physiological response. It involves defects in multiple organ systems - the muscle, the liver, the pancreas and the adipose tissue- act together to produce abnormal glucose and lipid metabolism.
Insulin resistance is a crucial factor for the development of type 2 diabetes and a major health problem for older adults. It is the principal mechanism by which obesity is considered to increase the risk for type 2 diabetes and is a key feature of the metabolic syndrome. The elevated prevalence of obesity and type 2 diabetes in the older population has important consequences on the morbidity and mortality as well as on the economic burden on our societies.
Controversy currently exists as to whether or not aging contributes to insulin resistance. Many potential factors confound the association between aging and insulin resistance, including obesity and physical activity. In addition, body types and specific lipid depositions may distinctly influence insulin resistance.
Pathophysiology of insulin resistance and type 2 diabetes
BODY COMPOSITION AND ECTOPIC LIPIDS
Body composition, meaning the amount of fat and muscle in the whole body also plays an important role in insulin resistance and metabolic diseases.
Due to its anatomical importance, skeletal muscle accounts for about 80% of the insulin stimulated glucose disposal. Sarcopenia consequences, such as frailty and mobility impairment, are implicated in falls, loss of independence and cost of care. In addition to preventive and therapeutic benefits, physical activity and exercise interventions have been proven to be cost-effective.
Specific adipose tissue depots distinctly influence insulin resistance, such as visceral adipose tissue and intermuscular fat accumulation. Ectopic lipid depositions, defined as an excess accumulation of triglycerides in non adipose tissues such as in the liver (intra-hepatic lipids, IHCL) and within the muscle fibers (intramyocellular lipids, IMCL), are positively associated with obesity and insulin resistance. Furthermore, the accumulation of intracellular lipids is often cited as being a key determinant in the underlying mechanisms of insulin resistance. In addition of playing an important role in obesity and type 2 diabetes, these ectopic fat depositions are also observed in common conditions such as aging and physical inactivity.
Our group uses in vivo and ex-vivo techniques to assess the different components of these fat depositions and how they relate to insulin resistance.
|
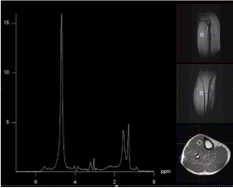
Example of spectra acquired with proton magnetic resonsance spectroscopy in a muscle of the lower leg.
|
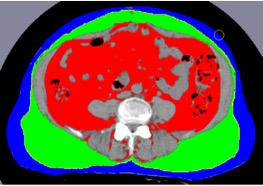
Example of the use of magnetic resonance imaging to quantify specific abdominal fat depositions.
|
EXERCISE EFFICIENCY AND METABOLIC FLEXIBILITY
In response to different environmental and physiological situations, different energy sources are used. A metabolically healthy individual is metabolically flexible and thus has the ability to switch from predominantly fat utilization to carbohydrate utilization when going from the fasted to the fed state. Obesity and type 2 diabetes blunt this capacity.
Different sources of energy are used during an acute bout of exercise depending on the intensity and the duration of the exercise bout, as well as on the environmental factors and the training state of the individual. Our focus is to continue to understand how to maximize metabolic flexibility and how this interacts with exercise efficiency in metabolically compromised individuals and in chronic diseases such as obesity and diabetes.
A volunteer undergoing a graded exercise test
FROM VOLUNTEERS TO CELLS TO MOLECULES: UNDERSTANDING MOLECULAR PATHWAYS IMPACTING MUSCLE METABOLISM
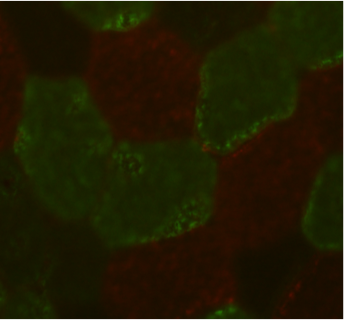
At the cellular level, substrate usage depends on multiple factors, among the principal ones are the mitochondria (power house of the cell). Mitochondria are cellular organelles that produce most of the chemical energy for cellular sustainment. In muscle, mitochondria are critically needed to generate energy for contraction and thus movement and locomotion. Mitochondria are highly adaptable; they respond to environmental challenges (such as exercise training or nutritional cues) by increasing or decreasing in numbers as well as in their content, and/or becoming more or less efficient.
 |
 |
|
Mitochondrial dysfunction is proposed to play a central role in the age-related physiological decline. At the cellular level, our focus is to investigate mitochondria functional adaptations through aging and exercise centering on pathways that impact mitochondria content, generation, dynamics and removal of damaged mitochondria (called mitophagy). |
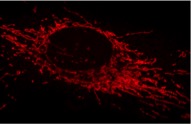
Mitochondrial network in one cell
|
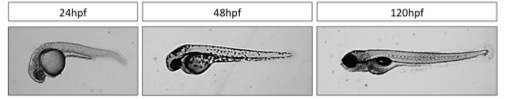
With the implementation of mechanistical models such as the use of zebrafish and cells, the lab is deepening research paradigms on specific pathways and protein targets.
Zebrafish, an ideal model for muscle development
Francesca Amati, Associate Professor
 |
After finishing her MD and specialization in internal medicine and diabetology at the University of Geneva, Francesca obtained her PhD at the University of Pittsburgh in the lab of Bret Goodpaster. The creation of the Aging and Muscle Metabolism lab started with the implementation of the AGIR study, a large clinical study including an exercise intervention. Progressively the lab has grown bonding clinical to biological approaches, thus creating a true translational environment. |
Sylviane Lagarrigue, Lab manager
 |
Sylviane studied biology at the University of Montpellier where she received her MSc. She did her PhD in the lab of Lluis Fajas Coll on the role of the CDK4 kinase in the biology of adipose tissue. Sylviane joined the AMM lab during for post doc growing her expertise in mitochondrial metabolism. As lab manager, Sylviane continues to develop new technical approaches and to lead scientific projects. |
Cassandra Tabasso, PhD student
 |
Cassandra studied biology at the University of Lausanne. After obtaining her MSc in biomedical sciences, she joined the AMM lab with the goal of investigating the link between insulin resistance and exercise. |
Axel Aguettaz, PhD student
 |
Axel obtained his Bachelor's degree in Biological Sciences at the University of Perugia, Italy. After obtaining a MSc in genetics in a double degree project between the University of Trieste and the University of Paris Diderot, he joined the AMM lab to investigate the role of perilipins in organelle contacts and exercise adaptation. |
Ammar Ebrahimi, Post doctoral fellow
 |
Ammar received his MSc in Medical Biotechnology from Tabriz University of Medical Sciences in Iran, where he worked on protein engineering. He obtained his PhD in Medical Biotechnology from Tehran University of Medical Sciences. During his PhD, he focused on lung tissue engineering and transplant immunomodulation. Following his PhD, he began working as a lecturer and researcher in the department of regenerative medicine at Guilan University of Medical Sciences, Iran. After joining the AMM lab, Ammar is developing mitophagy-inducing peptides. |
Mauricio Castro Sepulveda, Post doctoral fellow
 |
Strong of a training in movement sciences, Mauricio obtained his PhD in medical sciences at the Pontifica Universidad Catolica in Chile. His interest in human metabolism and mitochondrial dynamics in skeletal muscle brought him to join the AMM lab. Mauricio is developing his expertise in studying the effect of exercise in aging and particularly on the aging mitochondria. |
COLLABORATIONS & AFFILIATIONS
EXTERNAL SCIENTIFIC COLLABORATIONS
- Chris Boesch and Roland Kreis, Department of Clinical research, MR Spectroscopy and Methodology, University of Bern, Bern
- Cedric Moro, INSERM, Toulouse
- Johan Auwerx, EPFL, Lausanne
- Andrew Dwyer, Boston College, Boston
- Carles Cantó and Philipp Gut, Nestlé Institute of Health, Lausanne
- Anna Maria Cuervo and Susmita Kaushik, Einstein College of Medicine, New York
- Sarah Cohen, University of North Carolina, Chapel Hill
SCIENTIFIC COLLABORATIONS WITHIN THE UNIVERSITY OF LAUSANNE
- Jean Daraspe and Bruno Humbel, Electron microscopy facility
- Mehdi Tafti, Luc Pellerin and Christian Widman, Department of Physiology
- Zebrafish facility in collaboration with Alexandre Reymond (CIG) and Marie-Catherine Vozenin (CHUV)
CLINICAL COLLABORATIONS
- Vincent Gremeaux-Bader and Cyril Besson, Sports Medicine Lab, CHUV
- Didier Hans, Radiology, CHUV
- Leo Schluetter, Cardiologist, Private Practice
- Jean-Pierre Sacco, Let’s go Fitness
AFFILIATIONS
- Service of Endocrinology, Diabetology and Metabolism, CHUV
- Service of Sports Medicine, CHUV
- Sport research group/Plateforme de recherche sur le sport, Institute of Sports Sciences, UNIL
- Lausanne Integrative Metabolism Nutrition Alliance (LIMNA)
- Division of Endocrinology and Metabolism, University of Pittsburgh







Some articles/broadcasts about our research
RADIO
RTS la 1ère, le 12h30, Interview : Comment les athlètes se préparent-ils lors de fortes chaleurs?. July 9, 2023 (link)
RTS la 1ère, Le Grand Soir, June 2023 (link)
RTS la 1ère, CQFD, Les frileux en vedette, February 20, 2017 (link)
RTS la 1ère, CQFD, Il n’y a pas d’âge pour les bienfaits du sport, December 1, 2016 (link)
TV
Medscape TV, EASD 2022, Activité physique et diabète : quel que soit le type d’exercice, il faut bouger !. September 22, 2022 (link)
RSI, Falò, Invecchiare in forma, è possibile?. July 26, 2018 (link)
RTS, 36.9°, Vieillir Musclé. June 6, 2018 (link)
RTS, 36.9°, L’Amérique à vélo : l’incroyable défi d’une ex-sédentaire. April 11, 2018 (link)
TSR, 19h30, Le sport a des effets bénéfiques pour les seniors, December 1, 2016 (link)
WEB
Newsletter Echos du Vivant, November 2022 (link)
UNtoday, The official magazine of international civil servants, Prévenir, contrôler les effets délétères du diabète par l’activité physique. November 2022 (link)
La Repubblica, Salute, Il corpo degli atleti va forte? Tutto merito dei mitocondri. July 2021 (link)
Nature outlook, Sports sciences, Could mitochondria help athletes to make gains? March 2021 (link)
Coopération, Musclés par nature. March 26, 2019
In vivo magazine, supplément infographique In Extenso dédié au Pouvoir des muscles. November 2018
Sport Medicine Bulletin “Active Voice” March 2016 (link)
Sport Medicine Bulletin “Active Voice” June 2012 (link)
Men’s health magazine Online May 2012 (link)