Role of channel-activating proteases in epithelial ion transport and hypertension
Hypertension or increased blood pressure strongly associates with increased risk of cardiovascular events, stroke and kidney disease and affects one out of three adults in developed countries. Thereby, blood pressure is dependent on salt balance, and increase in the highly amiloride-sensitive epithelial sodium channel ENaC is a common phenomenon in salt-dependent hypertension.
The channel-activating proteases (CAPs) define a subclass of membrane-bound serine proteases. They induce ENaC-mediated Na+ ion transport through a novel autocrine mechanism. These serine proteases are further involved in multiple biological processes including embryonic development, barrier function of epithelia and organ differentiation, although their physiological function remains largely unknown. Recent findings indicate an important role in pathophysiological processes like hypertension and/or cancer.
Our laboratory focuses on two major goals:
-
-
- The study of channel-activating proteases in kidney physiology and pathophysiology like hypertension.
- The identification of downstream targets and underlying pathways of these proteases with focus on development of hypertension.
Adult mouse kidney -
kindly provided by Mariela Castelblanco Castelblanco
Using in silico, in vitro and in vivo genetic approaches, we like to understand the role of these serine proteases in physiological and pathophysiological conditions that provide targets for novel therapies in hypertension.
Keywords
Membrane-bound serine proteases, ENaC cleavage and activation, hypertension, genetic models
The kidney needs to adapt daily to variable dietary ion contents via various mechanism including diuretic, acid-base and hormonal changes.
Implication of channel-activating proteases (CAPs) in kidney function
Membrane-bound serine proteases, like the channel-activating proteases (CAPs) are suggested to regulate ENaC-mediated transport in the kidney, similar to what we previously demonstrated in lung (Planes et al., 2010) or colon (Malsure et al., 2014). Unexpectedly, when we tested the membrane serine protease CAP2/Tmprss4- deficient mice with respect to their Na+ balance through ENaC, we did not find any implication in kidney regulation (Keppner et al., 2015), rather we unveiled an important role in dysregulated renal water handling upon dietary potassium depletion (Keppner et al., 2019).
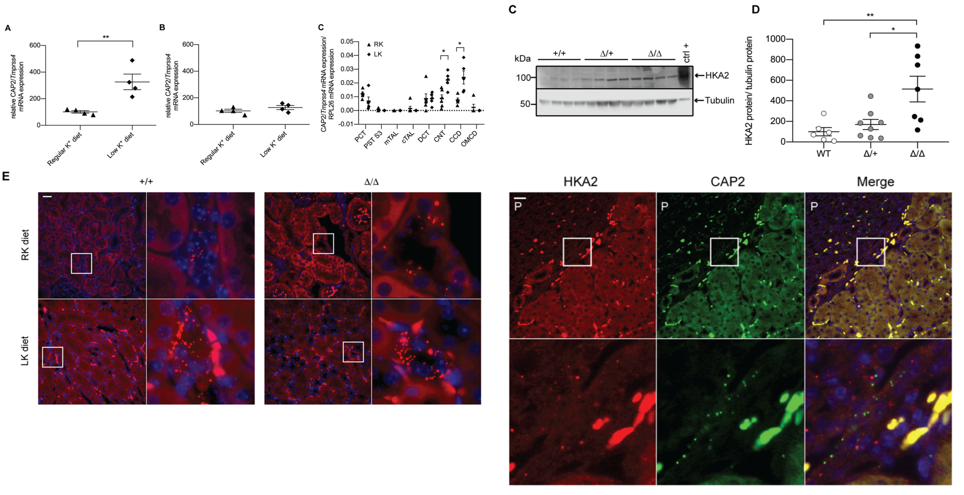
Keppner et al, Scientific Reports, 2019
Identification of downstream substrate targets of membrane-bound serine proteases
We will use proteomic and degradome data and include PICS (proteomic identification of protease cleavage sites) and protein-based TAILS (terminal amine isotopic labeling of substrates) in order to define the substrate specificity of these membrane-bound serine proteases and identify specific and common substrate targets under physiological and pathophysiological conditions.
We will further analyze whether the unveiled tissue- and/or context-specific regulation is dependent on protease cascade(s) and/or interaction of proteases with their inhibitors. This will help to clarify the role of these membrane-bound channel activating proteases in hypertension and to develop new therapies.
Edith Hummler, PhD, Associate Professor
 |
Edith Hummler was trained as biologist at the Universities of Tübingen and Göttingen, Germany, where she received her PhD. She performed a postdoctoral training with Günther Schütz at the German Cancer Research Center in Heidelberg, Germany, and then joined the group of Bernard Rossier at the Department of Pharmacology and Toxicology in Lausanne, Switzerland, and established her own research group using in vivo genetic engineering of models for human diseases of epithelial ion transport. Since 2016, she holds the position of an associate professor. Her research received funding from the Leducq Foundation Transatlantic Network in Hypertension, the Swiss National Science Foundation (SNSF), and the National Center of Competence in Research (NCCR): Kidney – control of homeostasis program of the SNSF.
Key words: epithelial ion transport, sodium and potassium homeostasis, channel-activating proteases, serine protease inhibitors, tissue remodeling. |
Muriel Auberson, Postdoc, lab manager
Elodie Boy, Postdoc
Sara Di Carlo, Postdoc
Emma Guignard, HES student
Mara Marangoni, Master student
Olivia Rickman, Postdoc
Paul-Emmanuel Vanderriele, Postdoc
Advanced search is available through Serval
Publications can be managed by accessing Serval via MyUnil
