JOYCE Lab
Our focus
Cancers develop in complex tissue environments, which they depend upon for sustained growth, invasion and metastasis. Inhibiting support from non-cancerous immune and stromal cells in the tumor microenvironment (TME) is considered to be an attractive therapeutic approach (Bejarano et al, 2021), as these cells are genetically normal and thus less likely to acquire drug resistance, a frequent outcome when targeting genomically unstable tumor cells. Different TMEs are populated by diverse cell types including innate and adaptive immune cells, fibroblasts, blood and lymphatic vascular networks, and specialized organ-specific cell types, in addition to the extracellular matrix, which collectively have critical functions in regulating tumorigenesis.
Our research program is focused on understanding how reciprocal communication between cancer cells and diverse immune and stromal cell types in the TME controls tumor initiation, progression, and metastasis, and modulates the response to therapeutic intervention. We employ a broad range of complementary experimental strategies to deeply interrogate the TME including the comprehensive analysis of patient samples (RNA-seq, proteomics, immune phenotyping, single cell analyses etc), 3D co-culture systems to mimic the TME, mouse models of cancer, multi-modal in vivo imaging platforms, and diverse computational analyses.
Our projects
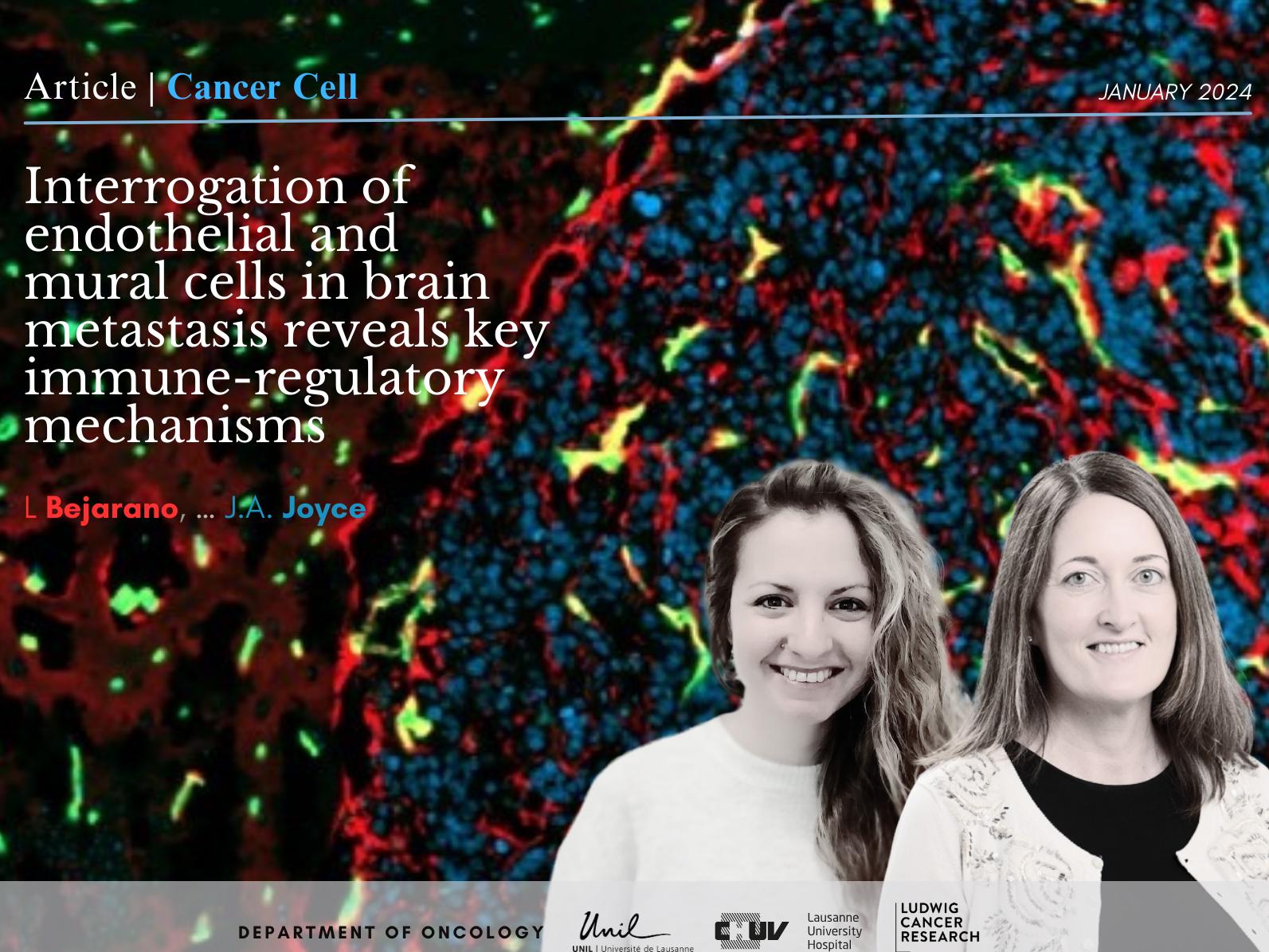
We currently have multiple projects focused on exploring the TME of primary and metastatic brain cancers, including analyses of diverse immune cell phenotypes, the blood and lymphatic vascular networks, and interrogation of the unique properties of the brain extracellular matrix. We are also fascinated by the dynamic evolution of the TME during cancer progression and metastasis in several tumor types, including breast and lung cancer. Another major area of interest centers on how different therapies alter the TME, and how those changes can consequently modulate therapeutic outcome. Our ultimate goal is to apply all of this knowledge to the clinic and develop targeted therapies that disrupt essential tumor-microenvironment interactions for the benefit of patients.
KEY PUBLICATIONS

- Bejarano L, Jordao MC, and Joyce JA (2021). Therapeutic targeting of the tumor microenvironment. Cancer Discovery 11: 933-959.
- Klemm F, Maas RR, Bowman RL, Kornete M, Soukup K, Nassiri S, Brouland JP, Iacobuzio-Donahue CA, Brennan C, Tabar V, Gutin PH, Daniel RT, Hegi ME, and Joyce JA (2020). Interrogation of the microenvironmental landscape in brain tumors reveals disease-specific alterations of immune cells. Cell 181: 1643-1660.
- Akkari L, Bowman RL, Tessier J, Klemm F, Handgraaf S, de Groot M, Quail DF, Tillard L, Gadiot J, Huse JT, Brandsma D, Westerga J, Watts C, and Joyce JA (2020). Dynamic changes in glioma macrophage populations after radiotherapy reveals CSF-1R inhibition as a strategy to overcome resistance. Science Translational Medicine 12(552):eaaw7843, doi: 10.1126/scitranslmed.aaw7843.
Meet all the Joyce Lab Members and Alumni.

| Funding |
|
| Awards |
Robert Bing Prize, Swiss Academy of Medical Sciences (2020); Cloëtta Prize (2018); Swiss Bridge Award (2017); American Cancer Society Scholar( 2012); Boyer Young Investigator Award (2011); Geoffrey Beene Junior Faculty Chair (2007); Rita Allen Foundation Scholar (2005); V Foundation Scholar (2005); Sidney Kimmel Foundation Scholar (2005). |
|
Affiliations |
|
| Links |





.png)


