PETROVA Lab
Our focus
Our main research interests are the mechanisms of tissue- and disease-specific functions of lymphatic and blood vessels. To study this question, our group combines expertise in advanced animal models with high resolution imaging, diverse cellular models and analyses of human patient samples.
Our projects
Mechanisms and novel effectors of (lymph)angiogenesis.
Lymphatic vasculature is a key player in the maintenance of normal fluid balance and immunity. It is also important in many common human diseases, such as cancer and chronic inflammatory conditions. Our aim is to identify central regulators of lymphatic vascular development and to study the impact of lymphatic vascular dysfunction on immunity and cancer. We employ tissue-specific inducible genetic animal models, ex vivo characterization of endothelial cells and analyses of patient samples.
Organ-/cancer-specific vasculature and its cross-talk with stem cells.
Blood-and-brain barrier and liver sinusoidal vasculature are examples of highly specialized vascular beds. Such specialization is essential for the normal organ function, for example to restrict the diffusion of certain components of blood to cerebrospinal fluid, and it is frequently affected in pathological conditions. However, with a notable exception of blood-and-brain barrier, our knowledge of the mechanisms underlying organ-specific vascular specialization and its role in human diseases is fragmentary. Even less is known about how such specialization affects the formation of primary or metastatic tumors, and whether it contributes to the maintenance of cancer stem cells. Using colon cancer as a model, we study stromal regulation of tissue- and cancer-specific stem cells. Our ultimate goal is to define new strategies for targeting stem cells in colon tumors.
Biomechanical forces in normal and diseased vessels.
Endothelial cells are continuously subjected to a variety of forces, such as shear fluid shear stress, stretch and transmural flow. In collaboration with bioengineers, we study how endothelial cells transmit and respond to biomechanical forces. Better understanding how biomechanical and biochemical stimuli are perceived and interpreted by vessels in normal and diseased tissues is important for targeting and regulation of vascular function.
KEY PUBLICATIONS

-
Petrova T.V., Koh G.Y. Biological functions of lymphatic vessels. Science, doi:10.1126/science.aax4063 (2020)
-
De Palma, M., Biziato, D., & Petrova. T,V. Microenvironmental regulation of tumour angiogenesis. Nat Rev Cancer, doi: 10.1038/nrc.2017.51 (2017).
-
Bernier-Latmani, J.,&Petrova, T.V. Intestinal lymphatic vessels: organization, mechanisms and functions Nat Rev Gastroenterol Hepatol, doi: 10.1038/nrgastro.2017.79. (2017)
Meet all the Petrova Lab Members.
|
Affiliations |
|
|
Links |
|
Lymphatic vessels and valves staining protocol
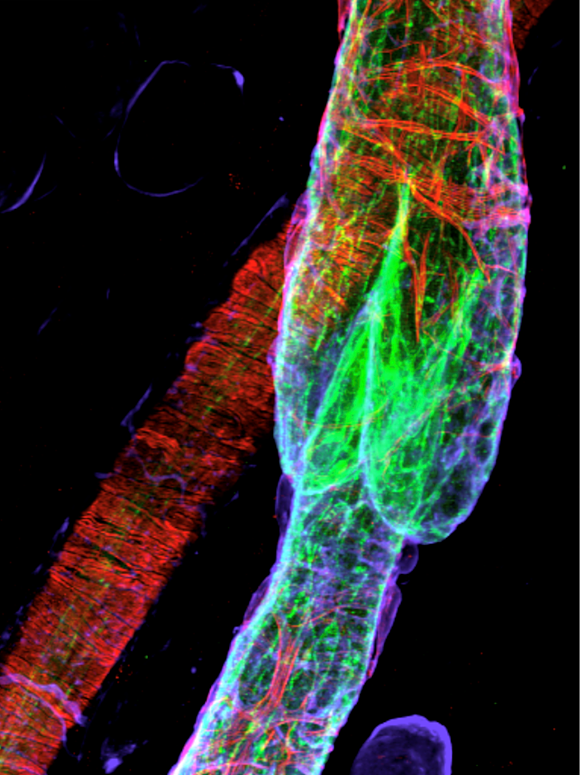
Lymphatic vessels transport interstitial fluid, immune cells, and antigens from tissues to lymph nodes. Unidirectional flow of lymph is maintained by lymphatic valves, complex and minute intraluminal structures, regularly distributed in lymphatic collecting vessels. Defects of lymphatic valves lead to fluid backflow and accumulation in tissues and impair tissue immunosurveillance. The rather small size (10-100 µm) and high cellular density of valves make their visualization tricky. The protocol by Sabine et al. describes the methods for valve characterization and quantification at the cellular level and approaches for studying lymphatic valve functionality.
Intestinal stroma staining and imaging protocol
Traditionally, histological analysis of the intestine was performed on paraffin or frozen thin sections. While this approach allows facile analysis of the gut epithelium, the finger-like structures of intestinal villi make stromal cell analysis difficult. Blood and lymphatic vessel analyses are best performed in 3D by wholemount immunostaining, but no protocol existed to easily visualize the intestine. We therefore developed the intestinal whole-mount staining protocol to perform quantitative analysis of intestinal blood and lymphatic vessels, while also allowing visualization and quantification of all intestinal cells.
Read the methodology paper
Single cell RNAseq of lymphatic endothelial cell subsets
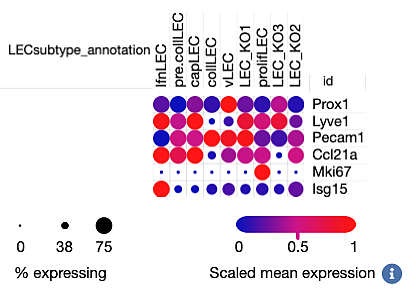
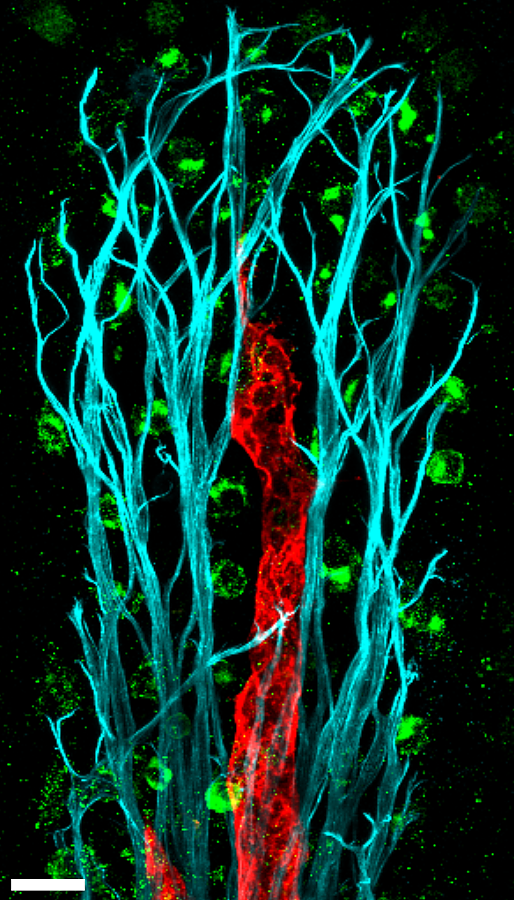
Lymphatic vascular tree is composed of capillaries, which take up fluid and cells from the interstitium, and pre-collecting and collecting lymphatic vessels, which transport lymph to lymph nodes. Collecting vessels harbor lymphatic valves, which function to prevent lymph backflow. The paper of Gonzalez-Loyola et al reports molecular characterization of mouse lymphatic endothelial subsets from these different compartments.
Explore the associated scRNAseq resource interactively here.
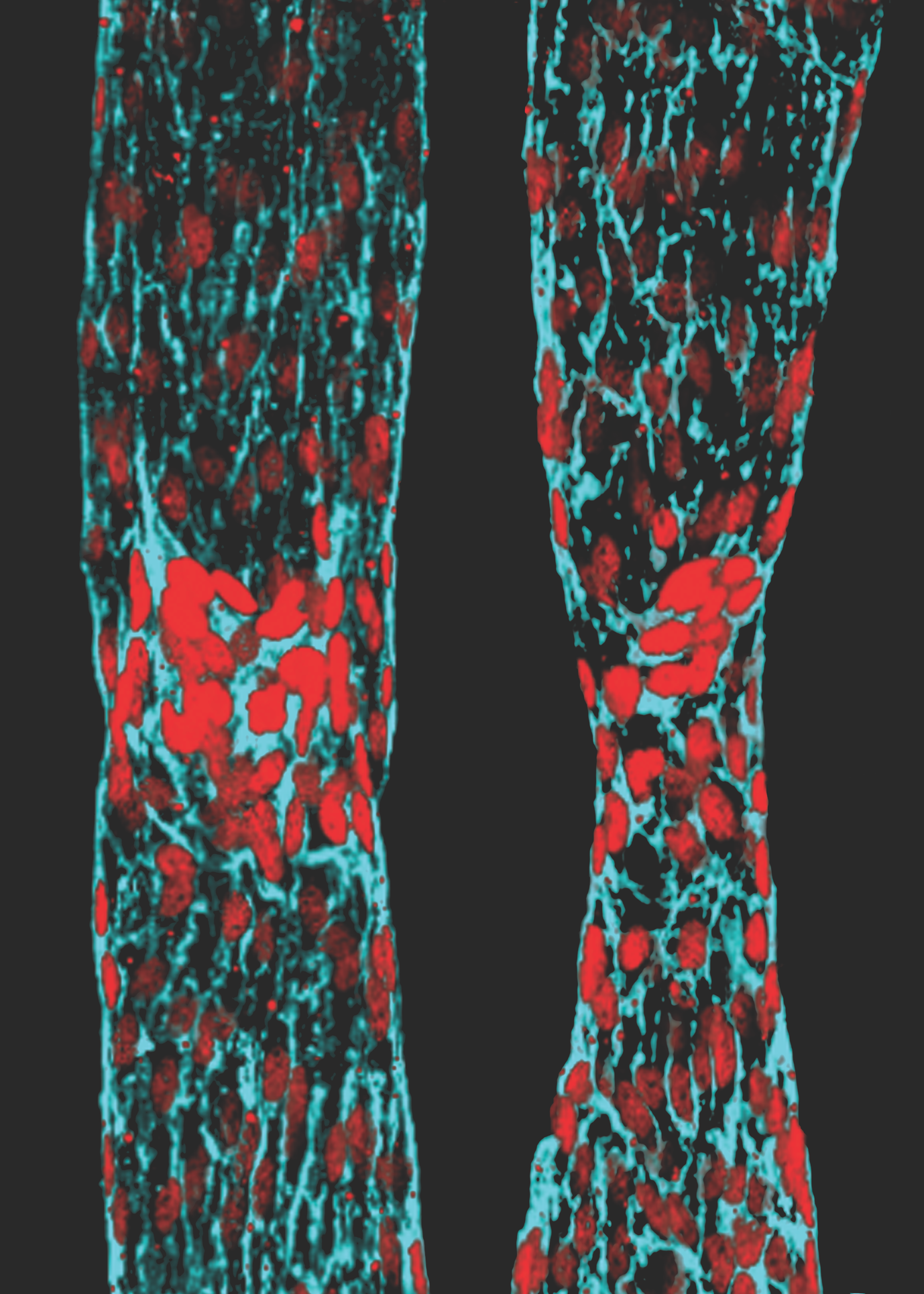
(c) Amélie Sabine






.png)


